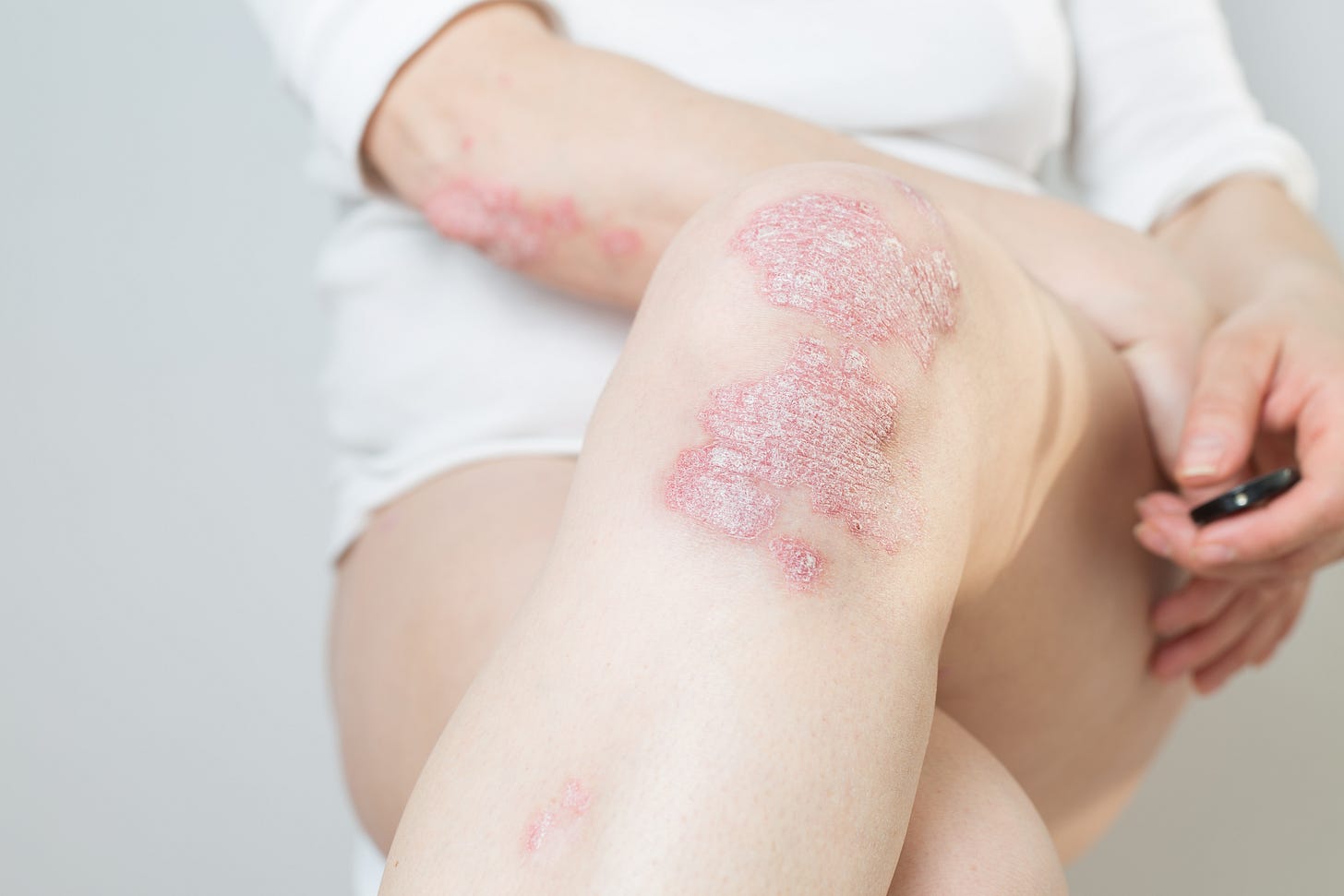
Bimekizumab: Pooled results from Phase 2 and Phase 3 randomized clinical trials
Today’s report also covers secukinumab dose optimization, lack of reactivation of latent tuberculosis, and more (1,300 words, 8 minutes 5 seconds)
The Vender on Psoriasis e-newsletter is supported by an unrestricted grant from Sun Pharma Canada
Good morning and welcome to the second issue of Special Reports: Skin Spectrum Weekly presents “Vender on Psoriasis.” This series is based on Dr. Vender’s popular column appearing in each edition of The Chronicle of Skin & Allergy, which offers expert commentary and opinions on current clinical developments in PsO. We’d love to get your feedback and suggestions and invite you to be in touch. Please write to us at health@chronicle.org

To evaluate the two-year safety profile of bimekizumab in patients with moderate to severe plaque psoriasis, researchers analyzed pooled results from four Phase 2 and four Phase 3 randomized clinical trials in patients who received at least one dose of bimekizumab. The study was published online in JAMA Dermatology. (doi:10.1001/jamadermatol.2022.1185 published online May 11, 2022).
A total of 1,789 patients (1,252 [70.0%] men; mean [SD] age, 45.2 [13.5] years) who had been treated with one or more doses of bimekizumab across the phase 2/3 trials were included. Total bimekizumab exposure was 3,109.7 person-years. Treatment-emergent adverse events (TEAEs), serious TEAEs, and TEAEs leading to treatment discontinuation were reported using exposure adjusted incidence rates (EAIRs) per 100 person-years.
According to the investigators, TEAEs occurred at an EAIR of 202.4 per 100 person-years and did not increase with longer duration of bimekizumab exposure. The three most frequently reported TEAEs were nasopharyngitis (19.1 per 100 person-years; 95%CI, 17.4-20.9 per 100 person-years), oral candidiasis (12.6 per 100 person-years; 95%CI, 11.3-14.0 per 100 person-years), and upper respiratory tract infection (8.9 per 100 person-years; 95%CI, 7.8-10.1 per 100 person-years). Most oral candidiasis events were considered mild or moderate; three events led to discontinuation. The EAIRs of inflammatory bowel disease (0.1 per 100 person-years; 95%CI, 0.0-0.3 per 100 person-years), adjudicated suicidal ideation and behaviour (0.0 per 100 person-years; 95%CI, 0.0-0.2 per 100 person-years), and adjudicated major adverse cardiac events (0.5 per 100 person-years; 95% CI, 0.3-0.8 per 100 person-years) were low.
Bimekizumab was well tolerated aside from an increased incidence of mild to moderate oral candidiasis in this pooled data from eight randomized clinical trials. Compared to previous reports, no safety signals were observed. Longer duration of treatment with bimekizumab did not increase risk of AEs.
Comment: Bimekizumab is a new anti-IL-17A and F blocker approved for the treatment of moderate to severe PsO. This is the fourth biologic against IL-17. It was found in clinical studies to be highly effective with superior PASI 90 responses with infrequent dosing of every four to eight weeks. This biologic has one safety concern that is higher than the other anti-IL17s: Candidiasis is likely the most common side effect to be seen with patients treated with bimekizumab. However, the incidence is very low. The majority of patients will NOT experience candidiasis. If it does occur, it is easily treatable without interruption of dosing and does not increase in frequency with continued dosing. The benefits still outweigh the minor risk presented by this safety concern.
Dose optimization of secukinumab in chronic plaque psoriasis
According to the results of this study published in Clinical and Experimental Dermatology (doi: 10.1111/ced.15157), monitoring secukinumab trough concentrations during maintenance therapy can help to identify patients who may benefit from treatment optimization since patients treated with secukinumab may show varying clinical responses. For example, weight-based dosing may be required to prevent underexposure in patients with higher body mass index (BMI).
In this multicentric, prospective study, the researchers worked to develop and validate a secukinumab immunoassay and potentially identify the therapeutic window in psoriasis patients. They collected secukinumab concentrations in sera from 78 patients with psoriasis at multiple timepoints at trough during the maintenance phase of treatment. Also, disease severity of each patient was assessed through the Psoriasis Area and Severity Index (PASI) during each visit to the hospital.
After quantification, 121 serum samples were included for exposure-response analysis with the secukinumab immunoassay that was developed. They noted that based on a linear mixed-effects model, secukinumab trough concentrations decreased with increasing BMI. Based on receiver operating characteristic (ROC) analysis, they determined that a minimal effective secukinumab threshold of 39.1 mg/L in steady state could be deduced and was associated with a 92.7% probability of having an optimal clinical response (PASI≤2 or ΔPASI≥90%).
Comment: This is a very interesting study with this commonly used anti-IL-17 biologic for moderate to severe psoriasis or psoriatic arthritis. Generally, the biomarker for skin psoriasis is the clinical visual exam of rapid and effective reduction of redness, thickness and scaling as well as itch. In psoriasis patients we never measure therapeutic levels of biologics and therefore this study is a new approach to potentially treating these patients with an adequate dose to either be able to titrate up dosing or reduce the dosage if clinical response is achieved. This may be more important in psoriatic arthritis however, and its use in dermatology is to be determined.
Lack of reactivation of latent tuberculosis in psoriasis patients treated with secukinumab
While some biologics for psoriasis, especially anti-tumour necrosis factor (TNF)-a therapies, may re-activate latent tuberculosis (TBC) infection, there is a low reported incidence of conversion to positive TBC status among patients with psoriasis treated with second-generation biologic therapies, particularly anti-interleukin (IL)-17 therapies such as secukinumab. However, in the clinical management of psoriasis, screening for tuberculosis infection is mandatory before starting biologic therapy.
According to protocols, psoriasis patients with a latent TBC infection with no evidence of clinically manifest active TB must undergo prophylactic treatment with isoniazid and/or rifampicin prior to the initiation of biologic therapy. This is also true for the newer biologics such as anti-interleukins (IL) 17 and 23, despite a low reported incidence of conversion to positive TBC status in patients treated with these therapies.
In this study published in the Journal of Dermatological Treatment (DOI:10.1080/09546634.2022.2062280), the safety profile of secukinumab in psoriasis patients with latent TBC infection was analyzed by real-life data. The data was collected using a retrospective chart review of patients with moderate-to severe psoriasis who showed positivity for TBC screening at baseline and then underwent secukinumab treatment for psoriasis at six Italian centres. Patients were treated with secukinumab 300 mg at weeks 0/1/2/3/4, then every fourweeks. A total of 59 patients were enrolled; 30.5% also had psoriatic arthritis and other comorbidities were common. At baseline, the mean psoriasis duration was 14.5 years. Ten (17%) patients did not undergo prophylaxis before starting secukinumab, but isoniazid ± rifampicin or rifampicin alone prophylaxis was administered in 49/59 (83.1%) patients.
Following a mean treatment duration of 84 weeks, the investigators noted there were no cases of TBC reactivation and no unexpected safety signals.
“Our study supports other analyses indicating that secukinumab may be used safely over an extended duration (up to 84 weeks) in psoriatic patients with latent TBC, even in patients who do not receive prior chemoprophylaxis,” the authors wrote, “and adds to the data supporting its use in patients with chronic inflammatory-mediated diseases. We recommend using IL-17A-targeted biologics as first-line therapy in psoriasis patients with latent TBC infection.”
Comment: Psoriasis patients are fortunate to have a wide variety of choices when choosing safe and effective biologics to treat their moderate to severe disease. The first studies of infliximab had the highest rates of latent TB conversions when the exact process of screening patients BEFORE taking this biologic was not yet established. Dermatologists quickly learned that screening for active and latent TB is important when starting anti-TNF biologics to prevent re-activation of this endemic respiratory disease that can be quite high in certain countries and populations. However, the newer biologics seem to still require testing for TB when it is unlikely to be necessary. This study shows that for secukinumab, this testing may not be required since there is no TB reactivation. However, one would caution that screening these patients prior to treating with any biologic with any MOA is good clinical practice. Sometimes, a switch to the TNF class of biologic may be suggested after failure or side effects from another class of biologic. The physician may have not realized the TB test may not have been performed because it was not needed for the previous biologic class. Doing the TB test for all patients seems to be part of standard of care presently for all biologic classes despite little or no evidence of problems.
If you find the contents of this newsletter interesting, please check out the Vender on Psoriasis podcast. It’s available at Apple iTunes, Stitcher, Spotify, or wherever you get your podcasts.
Dr. Ron Vender is a Hamilton-based certified dermatologist with 29 years of clinical practice experience and over 75 clinical trials in psoriasis. He is founder and director of Venderm Innovations in Psoriasis, a centre of excellence for Psoriasis offering a comprehensive management solution for individuals with psoriasis.
Upcoming events and learning opportunities
Registration is open for the 2022 Summer of Dialogue: Focus on Black Skin which will be held virtually on Wed. August 17, 7:00 PM to 8:30 PM EDT.
In recognition of persistent gaps in dermatologic training as it pertains to skin of colour, the Summer of Dialogue colloquium, Focus on Black Skin, will discuss recent progress on advancing inclusion of diverse skin tones in research and medical education.
This 90-minute live discussion will address treatment challenges specific to Black skin (Fitzpatrick Types V & VI), including phenotypic variations from standard training models and the necessity of cultural competence.
This non-accredited program will be available to attend at no charge thanks to the support of our sponsors.
Highlights of the event include:
The Dr. Mercy Alexis Keynote Lecture: Gaps in Aesthetic Treatment for Richly Pigmented Skin by Dr. Pearl Grimes of Los Angeles
Moderator: Dr. Andrew F. Alexis
Educating new and existing HCPs on optimal skin care for Black patients
Register for the 2022 Summer of Dialogue: Focus on Black Skin here: