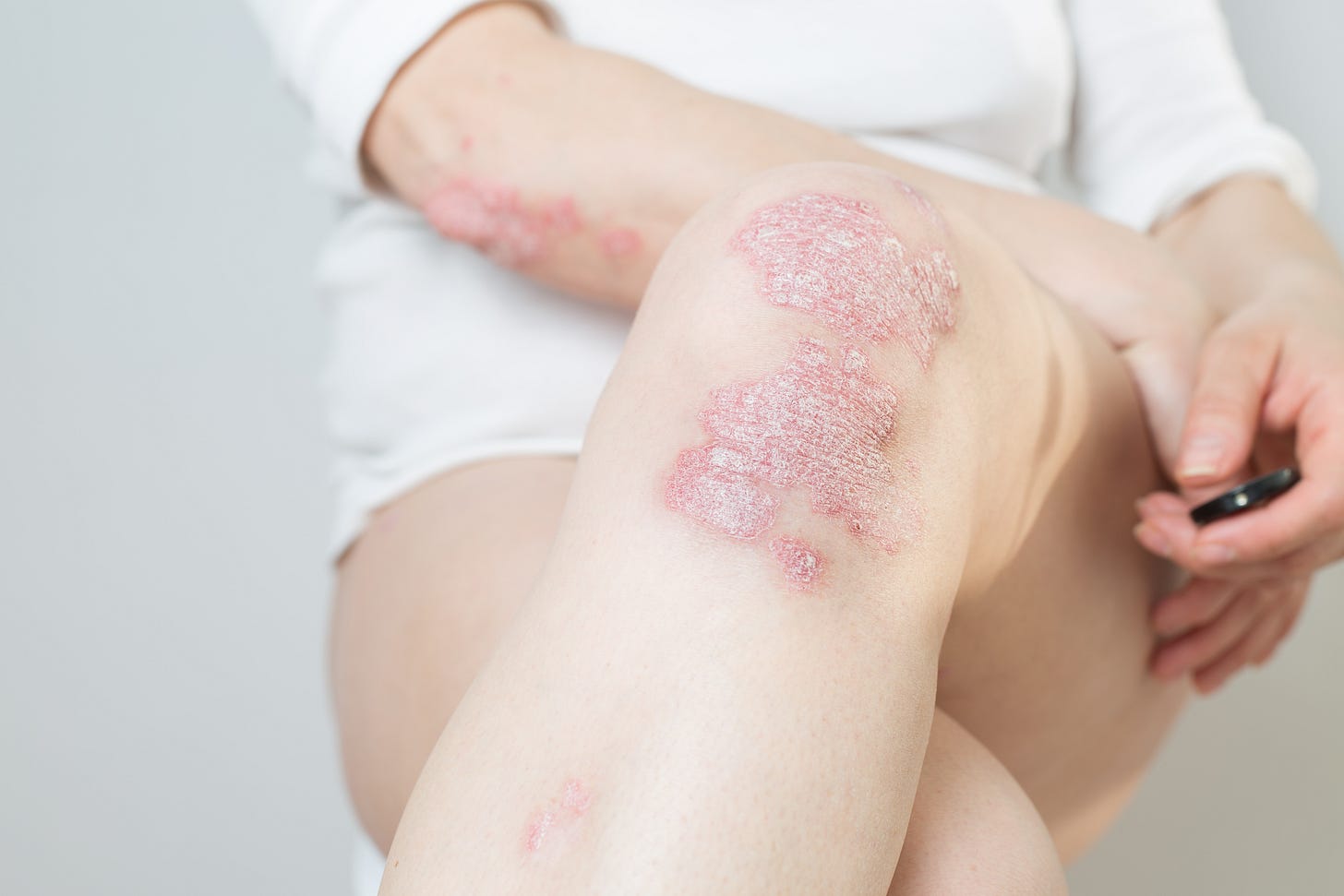
Deucravacitinib in Japanese patients with moderate to severe PP, GPP, or EP: A phase III trial
Today's report also covers efficacy and safety of piclidenoson, identification of psoriatic arthritis in patients with psoriasis, and more (1,900 words, 8 minutes, 30 seconds)
The Vender on Psoriasis e-newsletter is supported by an unrestricted grant from Sun Pharma Canada
Good morning and welcome to the eleventh issue of Special Reports: Skin Spectrum Weekly presents “Vender on Psoriasis.” This series is based on Dr. Vender’s popular column appearing in each edition of The Chronicle of Skin & Allergy, which offers expert commentary and opinions on current clinical developments in PsO. We’d love to get your feedback and suggestions and invite you to be in touch. Please write to us at health@chronicle.org

Deucravacitinib in Japanese patients with moderate to severe PP, GPP, or EP: A phase III trial
Deucravacitinib, an oral selective allosteric inhibitor of tyrosine kinase 2 (TYK2), has received approval in Japan for adult patients with plaque psoriasis (PP), generalized pustular psoriasis (GPP), and erythrodermic psoriasis (EP) who have not adequately responded to conventional systemic therapies. The approval stems from the outcomes observed in the global phase III POETYK PSO-1 and PSO-2 trials, which demonstrated improved efficacy compared to placebo in adults with moderate to severe plaque psoriasis. Additionally, findings from the POETYK PSO-4 trial, an open-label, single-arm, phase III study conducted in Japan, further support the efficacy and safety of deucravacitinib.
The POETYK PSO-4 trial in this study (J Dermatol. 2024; 1–15) involved 74 Japanese patients (76.2%, 66.7%, and 37.5% of patients with PP, GPP, and EP, respectively). The primary endpoints, including the proportion of patients achieving a ≥75% reduction from baseline in the Psoriasis Area and Severity Index (PASI 75) and a static Physician's Global Assessment score of 0 or 1 (sPGA 0/1) with at least a two-point improvement from baseline at week 16, were met. At week 16, substantial proportions of patients achieved PASI 75 and sPGA 0/1 across all psoriasis subtypes, with responses generally maintained through week 52.
Although adverse events (AEs) were common, with nasopharyngitis and acne being the most frequent, deucravacitinib was generally well-tolerated among Japanese patients. Rates of serious adverse events (SAEs) and discontinuations were low, and no deaths were reported. These findings underscore the effectiveness and safety of deucravacitinib over 52 weeks in Japanese patients with moderate to severe plaque psoriasis, and to a lesser extent, in those with GPP and EP. These results from the regional POETYK PSO-4 trial in Japan, in conjunction with the analysis of Japanese patients in the global POETYK PSO-1 trial, provide robust evidence supporting the efficacy and safety profile of deucravacitinib in the treatment of moderate to severe psoriasis in Japanese populations.
Comment: This study shows that deucravacitinib is certainly effective in a Japanese population. This population requires separate clinical trials to determine its efficacy and safety, due to the different genetic responses that can occur to different medications. The Japanese population is different than the rest of the world due to its specific diet high in fish and low-fat food. In addition, the mean weight of the Japanese psoriasis patient is generally lower than those in North America, or Europe. Pustular psoriasis is more common in the Japanese population and therefore finding medications that work specifically for pustular psoriasis or applying new indications to other medications that are not approved necessarily in North America for pustular psoriasis is important due to the increased incidence of this type of psoriasis that is very difficult to treat. Deucravacitinib has been shown to be helpful in this population. Side effects are extremely minimal. Whether or not this data could be extrapolated into a North American population is yet to be seen, however, since this would be off label use and not approved in the product monograph. Since there are no specific treatments approved for erythrodermic psoriasis one could consider deucravacitinib as a treatment option. However, the speed of response may not be as expected or as desired.
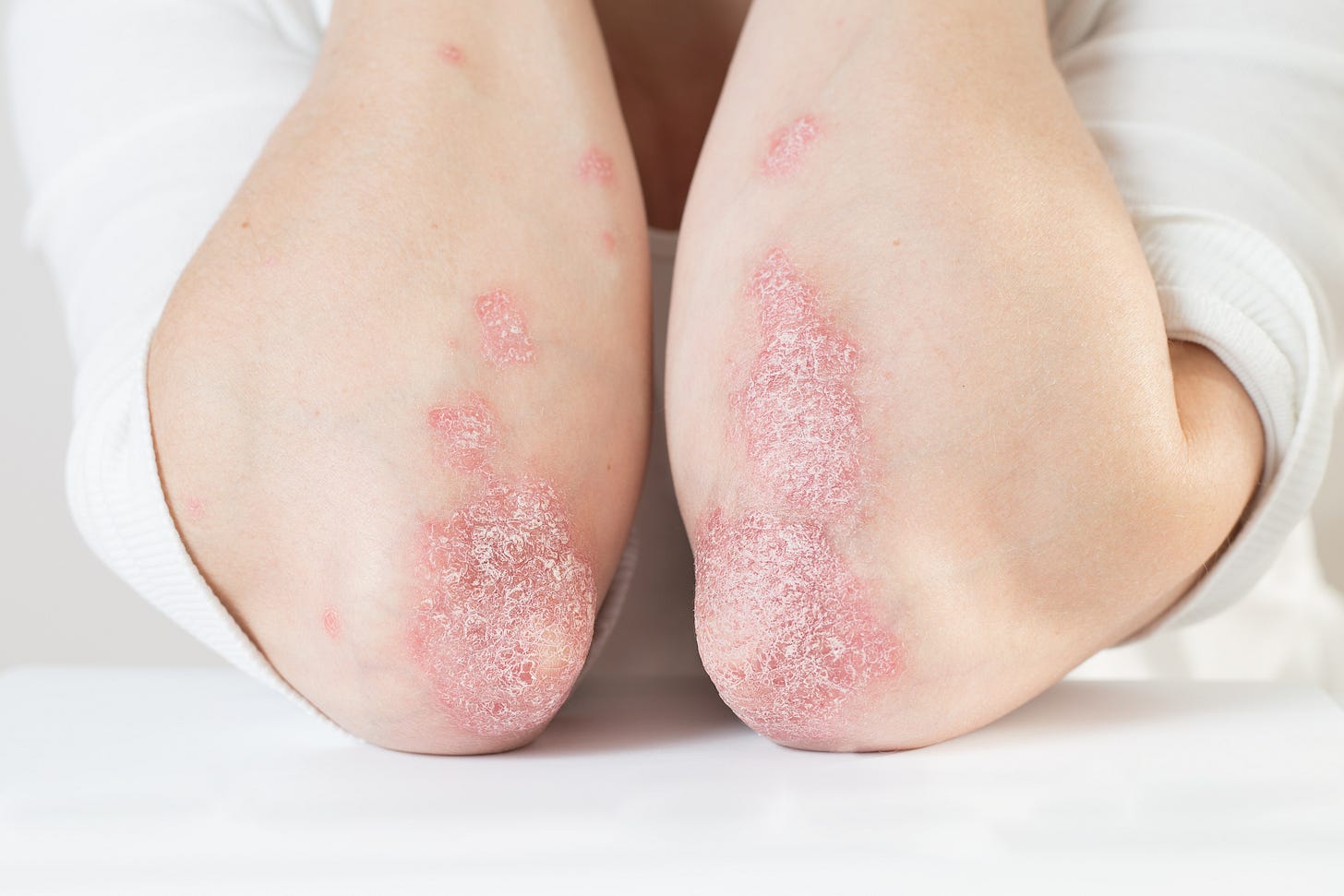
Efficacy and safety of piclidenoson in plaque psoriasis: COMFORT-1
The study aimed to evaluate the efficacy and safety of piclidenoson (CF101), an A3 adenosine receptor (A3AR) agonist, in treating moderate-to-severe plaque psoriasis. A3AR is known to be overexpressed in psoriasis patients, and piclidenoson has been shown to inhibit IL-17 and IL-23 production in keratinocytes, key cytokines involved in psoriasis pathogenesis.
The randomized, double-blind phase III COMFORT-1 trial involved 529 patients randomized into different treatment groups: piclidenoson 2 mg b.i.d., piclidenoson 3 mg b.i.d., apremilast 30 mg b.i.d., or placebo (J Eur Acad Dermatol Venereol 2024; 1–9). After 16 weeks, patients initially on placebo were re-randomized into active treatment arms. The primary endpoint was the proportion of patients achieving ≥75% improvement in Psoriasis Area and Severity Index (PASI-75) at Week 16 compared to placebo.
Results indicated that both doses of piclidenoson exhibited similar efficacy, with the 3 mg b.i.d. dose meeting the primary endpoint (PASI-75 rate of 9.7% versus 2.6% for piclidenoson versus placebo, p=0.037). The efficacy of piclidenoson continued to increase linearly throughout the study period. Furthermore, at week 32, a greater proportion of patients treated with piclidenoson 3 mg b.i.d. demonstrated improvement in Psoriasis Disability Index (PDI) compared to apremilast, with a trend towards significance (p=0.072). Piclidenoson also demonstrated a favourable safety profile compared to apremilast.
Piclidenoson showed increasing efficacy over time alongside excellent safety and tolerability. These findings support its further clinical development as a potential treatment for psoriasis.
Comment: New mechanisms of action are very interesting to investigate. It is also comforting to see that the specific interleukins associated with psoriasis, IL 17 and IL 23, are still the key cytokines responsible for the development and persistence of psoriasis. This new mechanism of action, A3AR, affects a different target, but in the end still is directed at reducing the interleukins mentioned above. Although this is an early study, it is promising to see that there is efficacy at least versus placebo and safety versus apremilast.
One important thing to note is that there seems to be a trend away from injectable treatments for psoriasis and back to the oral therapies. Oral therapies were once thought to be weaker than injectable biologics, but newer studies, such as this one, TYK2 inhibitors, oral peptides, and other mechanisms of actions are trending toward effective oral therapy. Patients are interested in oral therapy instead of injectables quite often when given a choice. However, at present there needs to be a balance between safety and efficacy which presently pushes clinicians to choose injectable biologic versus oral therapy. However, in the coming years, I believe we will see the trend change toward more effective oral therapies.
ID of PsA in patients with psoriasis
This article emphasizes the increased risk of psoriatic arthritis (PsA) in individuals with psoriasis (PsO) and the importance of early diagnosis (The Journal of Rheumatology 2023; 50:Suppl 2). Dermatologists are encouraged to screen patients with PsO for musculoskeletal symptoms and refer them to rheumatologists for diagnosis and treatment. Several screening questionnaires, such as the Psoriasis Epidemiology Screening Tool (PEST), the Psoriatic Arthritis Screening and Evaluation (PASE), the Toronto Psoriatic Arthritis Screen version 2 (ToPAS 2), and the Early Arthritis for Psoriatic Patients (EARP), have been developed to identify signs and symptoms of PsA. While these questionnaires have limited sensitivity and specificity, they can guide the need for a rheumatology assessment. Currently, there are no validated screening tools that require a physician’s assessment to confirm the presence of signs and symptoms suggestive of PsA, and no clinically applicable biomarkers available for screening.
Risk factors for incident PsA include severe PsO, psoriatic nail lesions, obesity, nonspecific musculoskeletal symptoms, and genetic factors. A study presented at the 2022 annual Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting developed risk prediction models for PsA in patients with PsO, which showed reasonable accuracy for predicting the development of PsA within one- and five-year time periods. Dermatologists are encouraged to annually screen their patients with PsO for incident PsA and to refer high-risk patients to rheumatologists. However, dermatologists face challenges in implementing screening, such as time constraints, competing priorities, and uncertainty about which diagnostic tools to use. The article also mentions that no single test can confirm a diagnosis of PsA, but imaging tests such as X-rays and MRI can help pinpoint changes in the joints that occur in PsA. The signs and symptoms of psoriatic arthritis often resemble those of rheumatoid arthritis, and both diseases cause joints to become painful, swollen, and warm to the touch. The article also discusses the prevalence of undiagnosed PsA among patients with psoriasis and the need for improved screening tools. The article highlights the importance of early diagnosis of PsA in patients with PsO, the development of risk prediction models for PsA, the challenges faced by dermatologists in implementing screening, and the need for improved screening tools.
Comment: Since psoriasis of the skin is usually the presenting symptoms of psoriatic disease and arthritis occurs later, it is important that the dermatologist be involved in screening for patients for psoriatic arthritis. There are numerous questionnaires, which are a favourite for rheumatologists. However, there is not one questionnaire that is perfect and allows a high specificity and sensitivity to determining whether a patient has psoriatic arthritis or not. Although it takes time to present the surveys to patients or takes up nursing staff or ancillary healthcare staff time, it is important for the dermatologist to ask simple questions such as “Do you have any joint pain?”
A simpler method was developed by Drs. Joe Merola and Alice Gottlieb, by asking PSA, which stands for P: Do you have any pain in any joints? S: Do you have joint stiffness in the morning or swelling of digits? A: Do you have any axial symptoms? This should take a matter of seconds, and could have a high yield in terms of referring to a rheumatologist. I do not feel it is the mandate of dermatologists to order X-rays or blood work, and should be left to the rheumatology experts. However, collaboration with a rheumatologist and multidisciplinary management of these patients is very beneficial and gives a great balance between improving skin and joint symptoms.
If you find the contents of this newsletter interesting, please check out the Vender on Psoriasis podcast. It’s available at Apple iTunes, Stitcher, Spotify, or wherever you get your podcasts.
Dr. Ron Vender is a Hamilton-based certified dermatologist with over 30 years of clinical practice experience and over 100 clinical trials in psoriasis. He is founder and director of Dermatrials Research Incorporated and Venderm Consulting, specializing in treatments and management solutions for individuals with psoriasis.
Subscribe to The Chronicle of Skin & Allergy
Established in 1995, The Chronicle of Skin & Allergy is a scientific newspaper providing news and information on practical therapeutics and clinical progress in dermatologic medicine.
To apply for a complimentary* subscription, please email health@chronicle.org with your contact information or click the link below.