Understanding psoriasis risk with immune checkpoint inhibitors
Today's report also covers novel oral inhibitors, real-world switching patterns, and more (1,800 words, 8 minutes, 10 seconds)
The Vender on Psoriasis e-newsletter is supported by an unrestricted grant from Sun Pharma Canada
Good morning and welcome to the 21st issue of Special Reports: Skin Spectrum Weekly presents “Vender on Psoriasis.” This series is based on Dr. Vender’s popular column appearing in each edition of The Chronicle of Skin & Allergy, which offers expert commentary and opinions on current clinical developments in PsO. We’d love to get your feedback and suggestions and invite you to be in touch. Please write to us at health@chronicle.org

Understanding psoriasis risk with immune checkpoint inhibitors
This cohort study investigated the association between immune checkpoint inhibitor (ICI) therapy and the risk of psoriasis in cancer patients (JAMA Dermatol 2025; 161(1):31-38.). The researchers utilized data from the Taiwan National Health Insurance database and the Taiwan Cancer Registry, employing a target trial emulation framework to minimize common biases.
The study included 135,230 patients who received antineoplastic medications between Jan. 2019 and June 2021. Of these, 3,188 were treated with ICIs (PD-1 and PD-L1 inhibitors), while 132,042 received chemotherapy or targeted therapies. The mean follow-up period was 1.46 years.
The researchers found that ICI users experienced a higher incidence of psoriasis (5.76 cases per 1,000 person-years) compared to non-ICI users (1.44 cases per 1,000 person-years). After adjusting for demographics, comorbidities, and outpatient visits, ICI users showed a significantly increased risk of developing psoriasis: Cox proportional hazards model: IPTW-adjusted HR of 3.31; Fine-Gray subdistribution hazard model: IPTW-adjusted SHR of 2.43.
They noted the risk of developing psoriasis was highest within the first 180 days of ICI initiation (IPTW-adjusted SHR, 7.69). Subgroup analyses revealed statistically significant higher risks among ICI users aged 66 years and older, as well as male patients.
Comparison with other anticancer therapies showed that ICI users had the highest risk of psoriasis, followed by protease kinase inhibitor users, with chemotherapy and monoclonal antibody users showing the least risk.
The study’s strengths include its large-scale, nationwide design and the use of a simulated target trial framework to minimize biases. However, limitations include the lack of data on lifestyle factors, genetic predisposition, and psoriasis severity.
The authors propose that the increased risk of psoriasis in ICI users may be related to the shared biological processes between psoriasis and ICI-induced immune-related adverse events. ICIs block inhibitory pathways such as PD-1/PD-L1, leading to T cell reactivation and potentially enhanced immune activity, which may manifest as psoriasis.
This study demonstrates a higher risk of psoriasis in cancer patients receiving ICI therapy compared to those on chemotherapy or targeted therapy. While this adverse effect is relatively uncommon, the findings highlight the importance of awareness among medical professionals to ensure optimal cancer care and skin health management in patients undergoing ICI treatment.
Comment: This study offers an analysis of the increased risk of psoriasis among cancer patients treated with immune checkpoint inhibitors (ICIs). The use of a large, nationwide cohort and target trial reduces selection bias, strengthening its validity. The finding that ICI users have over three times the risk of developing psoriasis compared to non-ICI users underscores the immunomodulatory effects of PD-1/PD-L1 inhibition. However, limitations include the lack of genetic and lifestyle data, which can influence psoriasis risk. Clinically, this study highlights the importance of derm monitoring in patients receiving ICIs, especially in older men who appear at higher risk.
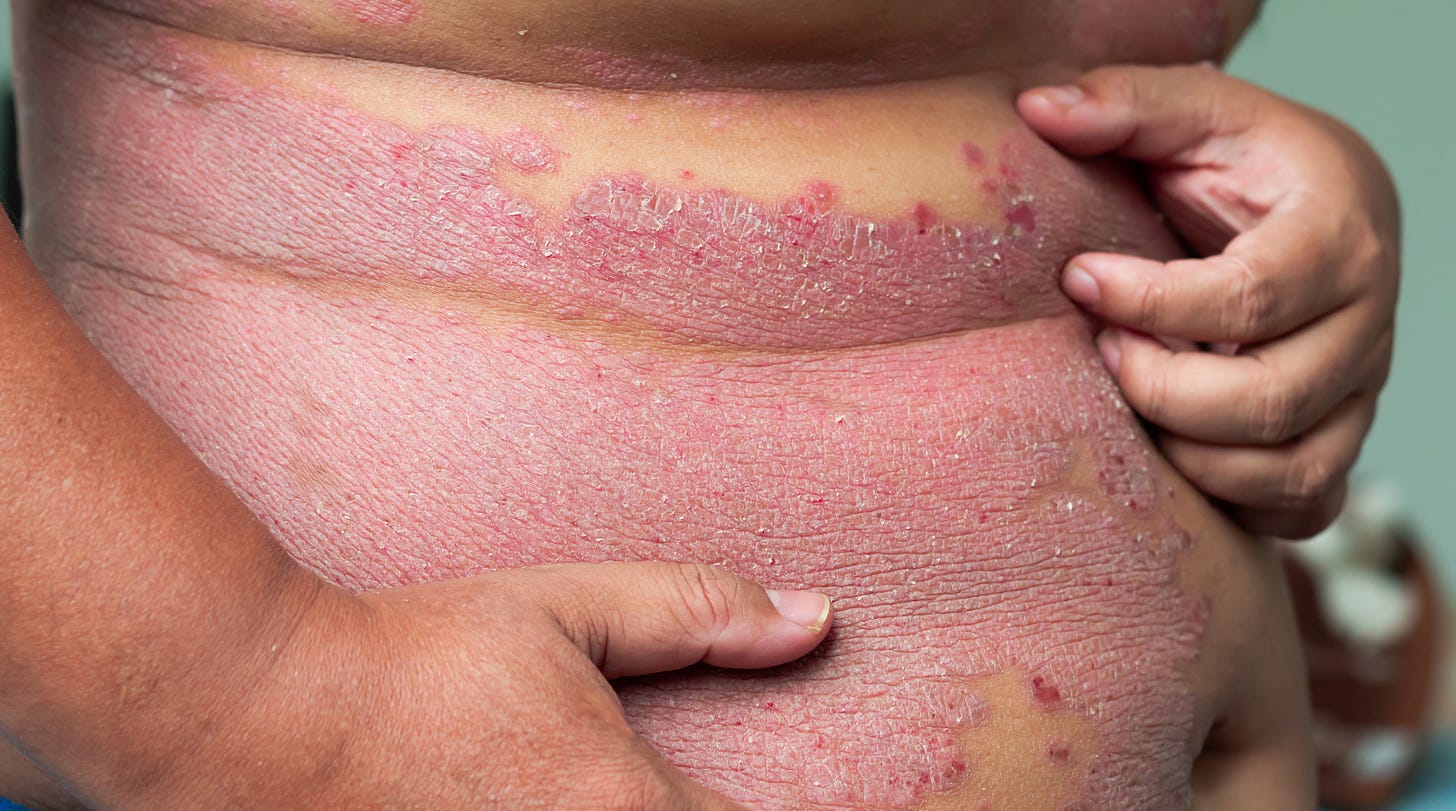
A Phase II randomized trial of a novel oral RORγt inhibitor for moderate-to-severe plaque psoriasis
This paper reports on a Phase II randomized trial evaluating BI 730357, a novel oral RORγt inhibitor, for the treatment of moderate-to-severe plaque psoriasis (The Journal of Investigative Dermatology 2025). The study consisted of two parts and a long-term extension (LTE) trial.
In Part 1, 178 patients were randomized to receive BI 730357 at doses of 25, 50, 100, or 200 mg once daily (QD), or placebo under fasting conditions for 12 weeks. The co-primary endpoints were achievement of PASI 75 (≥75% reduction from baseline in Psoriasis Area Severity Index) and sPGA 0/1 (static physician’s global assessment score of clear/almost clear) at week 12. At week 12, 30.0% of patients in the 200 mg QD group achieved PASI 75 (p=0.0062 vs placebo) and 27.5% achieved sPGA 0/1 (p=0.0095 vs placebo).
Part 2 enrolled 96 patients who were randomized to BI 730357 400 mg QD, 200 mg twice daily (BID), or placebo under fed conditions. At week 12, 25.6% and 23.8% of patients in the 400 mg QD and 200 mg BID groups, respectively, achieved both PASI 75 and sPGA 0/1.
An exposure-response analysis revealed that the change from baseline in PASI score plateaued at higher BI 730357 exposures, with a maximum effect of approximately 50% relative change from baseline at week 12. This indicated no notable increase in efficacy for doses ≥200 mg QD.
The LTE trial included 165 patients from Parts 1 and 2. Over the course of treatment, 56.4% of patients achieved PASI 75, though 26.9% of these responders lost their response by the last efficacy assessment.
Safety data showed that ≤15.8% of patients experienced drug-related adverse events (AEs) in Trial 1407-0030. The most common AEs were infections and infestations, particularly upper respiratory tract infections and nasopharyngitis. Serious AEs were infrequent and generally not considered drug-related.
The authors conclude that while RORγt inhibition with BI 730357 led to meaningful decreases in disease activity for some patients, the extent of disease control was limited. The efficacy plateau observed at higher drug exposures suggests that the relationship between RORγt and IL-17 may be more complex than initially thought. The authors propose that these findings warrant a re-evaluation of the central role of RORγt in IL-17 stimulation and the mechanism of action of targeting a transcription factor to inhibit Th17 differentiation and IL-17 production.
BI 730357 demonstrated moderate efficacy and was well-tolerated in patients with moderate-to-severe plaque psoriasis. However, the limited extent of disease control and the unexpected efficacy plateau at higher doses highlight the need for further research to fully understand the RORγt/IL-17 pathway in psoriasis pathogenesis and treatment.
Comment: This Phase II trial provides new information into RORγt inhibition as a therapeutic approach for psoriasis. While BI 730357 demonstrated moderate efficacy, the unexpected plateau in PASI response at higher doses raises questions about the mechanism of RORγt targeting. The study design, including an exposure-response analysis and long-term extension data, is a strength. However, the observed treatment effect remains modest, and the long-term durability of responses is uncertain. The findings suggest that RORγt may not be as central to IL-17 regulation in psoriasis as previously assumed, needing further studies before moving on to Phase III trials.

Real-world switching patterns and associated characteristics in patients with psoriasis treated with biologics
This retrospective cohort study, conducted in Greece, analyzed real-world switching patterns and associated characteristics in patients with psoriasis treated with biologics (Archives of Dermatological Research 2025; 317:310).
The study utilized data from the Greek digital prescription database, including 6,772 biologic-naïve patients who initiated treatment between Jan. 1, 2016, and Dec. 31, 2020.
The results revealed several key findings regarding switching rates and patterns. The overall switching rate from first to second biologic treatment was 12.4% (Scenario 1) and 13.4% (Scenario 2). Anti-TNFα agents demonstrated higher switching frequencies compared to interleukin-targeting antibodies (20.9% vs. 9.8%). Notably, inter-class switching from anti-TNFα to anti-interleukin agents was 3.3 times higher than the opposite transition. Among the specific drugs, brodalumab, ustekinumab, and secukinumab showed the lowest switching rates, while infliximab had the highest at 44%. Interestingly, ustekinumab and secukinumab had higher re-initiation rates than switches.
The study also examined the time to switch or re-initiation. The median time from first to second line was 43.3 months. Patients receiving brodalumab and certolizumab pegol did not reach median time to switch, while among those reaching median time, secukinumab and ustekinumab had the highest.
The study employed two scenarios in its methodology. Scenario 1 considered both treatment switches and re-initiations after a 90-day gap, while Scenario 2 focused solely on switches to different biologics. Statistical analyses included Kaplan-Meier curves, cumulative incidence functions, and Cox regression models.
These findings have several implications for clinical practice. The significantly reduced switching risk of interleukin-targeting antibodies compared to TNFαagents suggests better treatment persistence. The higher switching rates from anti-TNFα to interleukin-targeting treatments indicate a potential superiority of newer biologics in maintaining treatment response. The study also highlights the importance of considering patient characteristics, such as gender and presence of PsA, when selecting and managing biologic treatments for psoriasis. These insights provide valuable guidance for clinicians in selecting appropriate biologic agents and managing psoriasis treatment in real-world settings.
However, the study lacks clinical details on reasons for treatment switching or re-initiation, preventing conclusions on efficacy or safety as potential drivers of therapy modifications. The inability to assess the impact of evolving comorbidities or lifestyle changes on switching patterns is another limitation. Due to its retrospective nature, the study may have potential selection bias. Additionally, the study is limited to a specific time period, which may not capture long-term trends in biologic use.
Comment: This retrospective cohort study contributes valuable real-world data on biologic therapy persistence and switching patterns in psoriasis. The lower switching rates for IL-17 and IL-23 inhibitors compared to TNF-α inhibitors reinforce their superior long-term efficacy. The higher likelihood of switching from TNF-α to interleukin-targeting agents aligns with current clinical practice favouring newer biologics. The study is limited by the absence of clinical reasons for switching, preventing conclusions about efficacy versus safety concerns. However, the reason for switch is often primary or secondary loss of response. Despite these limitations, the findings offer practical insights into biologic treatment optimization and patient management in dermatology practice applicable to Canadian derm practices.
If you find the contents of this newsletter interesting, please check out the Vender on Psoriasis podcast. It’s available at Apple iTunes, Stitcher, Spotify, or wherever you get your podcasts.
Dr. Ron Vender is a Hamilton-based certified dermatologist with over 30 years of clinical practice experience and over 100 clinical trials in psoriasis. He is founder and director of Dermatrials Research Incorporated and Venderm Consulting, specializing in treatments and management solutions for individuals with psoriasis.
Subscribe to The Chronicle of Skin & Allergy
Established in 1995, The Chronicle of Skin & Allergy is a scientific newspaper providing news and information on practical therapeutics and clinical progress in dermatologic medicine.
To apply for a complimentary* subscription, please email health@chronicle.org with your contact information or click the link below.




